|
|
|
|
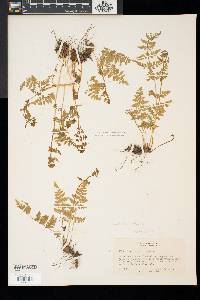
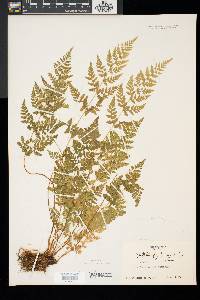
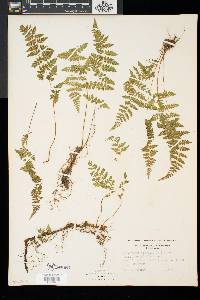
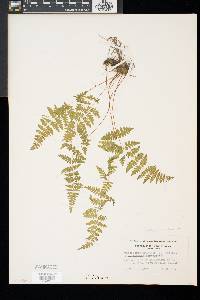
Family: Cystopteridaceae
[Cystopteris filix-fragilis (L.) Chiov.] |
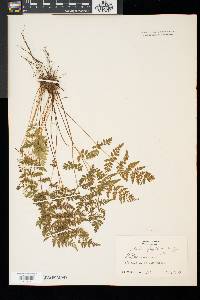
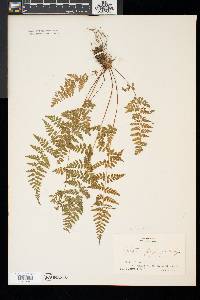
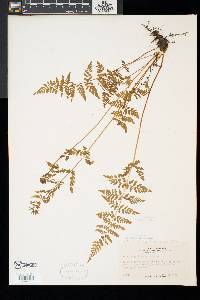
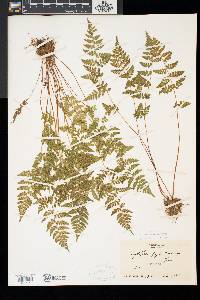
Stems creeping, not cordlike, internodes short, beset with old petiole bases, hairs absent; scales tan to light brown, lanceolate, radial walls thin, luminae tan. Leaves monomorphic, clustered at stem apex, to 40 cm, nearly all bearing sori. Petiole dark at base, mostly green to straw-colored distally, shorter than or nearly equaling blade, base sparsely scaly. Blade lanceolate to narrowly elliptic, 1(--2)-pinnate-pinnatifid, widest at or just below middle, apex acute; rachis and costae lacking gland-tipped hairs or bulblets; axils of pinnae lacking multicellular, gland-tipped hairs. Pinnae usually perpendicular to rachis, not curving toward blade apex, margins serrate to sharply dentate; proximal pinnae pinnatifid to pinnate-pinnatifid, ± equilateral, basiscopic pinnules not enlarged; basal basiscopic pinnules sessile, base truncate to obtuse, distal pinnae deltate to ovate. Veins directed mostly into teeth. Indusia ovate to lanceolate, without gland-tipped hairs. Spores spiny or verrucate, usually 39--60 µm. 2 n = 168, 252. Sporulating summer--fall. Mostly on cliff faces, also in thin soil over rock; 0--4500 m; Greenland; St. Pierre and Miquelon; Alta., B.C., Man., N.B., Nfld., N.W.T., N.S., Ont., P.E.I., Que., Sask., Yukon; Alaska, Calif., Colo., Conn., Idaho, Ill., Ind., Iowa, Kans., Maine, Mass., Mich., Minn., Mont., Nebr., Nev., N.H., N.Mex., N.Y., N.Dak., Ohio, Oreg., Pa., S.Dak., Tex., Utah, Vt., Wash., Wis., Wyo.; worldwide. Cystopteris fragilis is most often confused with C . tenuis in the east and C . reevesiana in the southwest. Habitat and geography, as well as the morphologic features discussed in the key, usually serve to separate these taxa. For instance, C . fragilis is more likely to be found on cliffs whereas the other species prefer boulders and soil ( C . fragilis occurs at higher elevations and/or latitudes than the other species). These distinctions can be confounded when C . fragilis forms hybrids with sympatric species. Sterile pentaploid plants have been discovered where C . fragilis overlaps with C . laurentiana , tetraploid hybrids are likely where C . fragilis occurs with C . tenuis , and triploids may form where C . fragilis is found with C . reevesiana . Even after segregating relatively distinct elements such as Cystopteris protrusa , C . reevesiana , and C . tenuis , and identifying sterile hybrids, C . fragilis still remains a polymorphic and complex taxon that probably contains a number of natural, cryptic evolutionary units. For example, morphologically distinct hexaploid cytotypes have been reported (C. H. Haufler and M. D. Windham 1991). These occur as isolated and disjunct populations in Ontario, Alaska, Arizona, Colorado, Montana, and Wyoming. Isozymic profiles of each of these populations indicate that the hexaploids are polyphyletic and should not be accorded species status. The presence of verrucate spores (as opposed to the normal spiny spores) has been used to circumscribe Cystopteris dickieana . Although genetic analyses have not been undertaken, we think the verrucate spore is probably a recessive feature controlled by one or a few genes. While present at low frequency in much of the range of C . fragilis , verrucate spores are particularly prominent in the Great Plains. Perhaps in this region the genetic combinations specifying verrucate spores have been fixed. Following R. F. Blasdell (1963), C . dickieana is also considered here to be conspecific with C . fragilis because (1) early stages in the development of spiny spores can appear verrucate (A. C. Jermy and L. Harper 1971), (2) the hexaploid cytotypes discussed above always have verrucate spores, regardless of their parentage, (3) individuals with verrucate spores can be found in populations that are otherwise uniformly spiny-spored, and (4) individuals and populations that have verrucate spores are not otherwise (morphologically, ecologically, or genetically) distinct from those that have spiny spores. Especially in the western portion of its North American range (British Columbia, Washington, Montana, Idaho, Oregon, California), Cystopteris fragilis appears to be developing morphologically and ecologically distinctive variants. Hybrid individuals with aborted spores have been discovered, and plants from these areas increasingly tend to grow on both soil and rock and to have slightly different morphologies on the two substrates. These variants intergrade, however, and are not sufficiently distinct to warrant species status. This polymorphic polyploid is probably actively speciating at the tetraploid level, perhaps through gene silencing (C. R. Werth and M. D. Windham 1991).
Lvs glabrous or inconspicuously glandular, in a small cluster on a short, densely scaly rhizome, 0.5-3.5(-4) dm, the dark brown (or distally greenish) petiole shorter than or to as long as the blade; blade mostly 3.5-20(-25) נ1.5-8 cm, (2-)2.5-4(-5) times as long as wide, 2-3 times pinnate, with 8-18 pairs of pinnae (the members of a pair often offset, especially the distal pairs), the lowest pair equaling or generally a little smaller than the next pair, the upper ones gradually reduced so that the blade tapers into a narrow tip; veins typically directed to the teeth of the pinnules, but the tooth sometimes apically slightly indented so that the vein ends in a small sinus; indusium pale, not glandular; 2n=188 in ours. Moist, mostly wooded slopes and ledges in circumneutral soil; circumboreal, s. in our range to Va. and Mo. We have 2 vars. with widely overlapping distribution: Gleason, Henry A. & Cronquist, Arthur J. 1991. Manual of vascular plants of northeastern United States and adjacent Canada. lxxv + 910 pp. ©The New York Botanical Garden. All rights reserved. Used by permission. |
The PCC, and this data portal, were made possible by funding from the National Science Foundation’s
Advancing Digitization of Biological Collections (ADBC) program, grant numbers
1802504,
1802352,
1802134,
1802033,
1802270,
1802255,
1802239,
1802446,
1802305.
The pteridoportal taxonomic thesaurus is based on the
Checklist of Ferns and Lycophytes of the World, generously provided by Michael Hassler.